
- WHAT IS ISO 13485 IN PLAIN ENGLISH HOW TO
- WHAT IS ISO 13485 IN PLAIN ENGLISH UPDATE
- WHAT IS ISO 13485 IN PLAIN ENGLISH PROFESSIONAL
WHAT IS ISO 13485 IN PLAIN ENGLISH HOW TO
No matter if you are new or experienced in the field, this book gives you everything you will ever need to learn on how to handle ISO 9001, ISO 14001, ISO 27001, ISO 22301, ISO 20000, ISO 22000, OHSAS 18001, ISO 13485, AS9100, and IATF 16949 documents. If you would like a quote or would need to ask questions, please contact us.In this book, Dejan Kosutic, author and experienced ISO consultant, is giving away his practical know-how on managing policies, procedures, plans, forms, reports, and other documented information. You’ll want to know about any deficiencies or gaps in the system before a regulatory auditor arrives!ĭepending on the complexity of your operations, our recommendation is to aim between 6 to 12 months to achieve ISO 13485 registration. It is also worth considering having a compliance audit performed by a third party when you’ve finished your QMS to see if everything has been covered. We can customise and make the templates easy to use and also train your staff in document writing so that in the future your QMS documents look like a consistent set of documents instead of a collection of disparate files (a common problem as well as a red flag to an auditor!). In this regard, if you are short on time and resources, we are also able to provide consultants and document writers to your site to assist. Based on the modest cost, the templates are well worth investing in, however, a template pack will never work straight out of the box and they will require a degree of specification and customisation to detail how your business, facility and quality processes should work. PharmOut sells QMS templates which will help to set you up with good Quality foundation on which you can structure the rest of your company.
WHAT IS ISO 13485 IN PLAIN ENGLISH PROFESSIONAL
In an ideal situation, the people in your organisation who own the processes and are subject matter experts should write the documents – however, in reality, often they don’t have the time or technical writing background to do it! PharmOut’s professional document writers will work closely with your team to extract the crucial information and translate it into effective and compliant documents. Document management and change control are frequent sources of non-compliance especially if the system has been set up in a haphazard way – an easy target for auditors! With this in mind, if you have more than 50 documents in your current QMS (or fewer but are looking to expand) it is paramount to invest in a document management system and ensure you have an effective change control system in place. The majority of changes in regulated companies will be to quality documents. Our Recommendations for a Successful QMS Implementation Here are some key points to consider when implementing a QMS in regulated company or a Quality Critical facility:

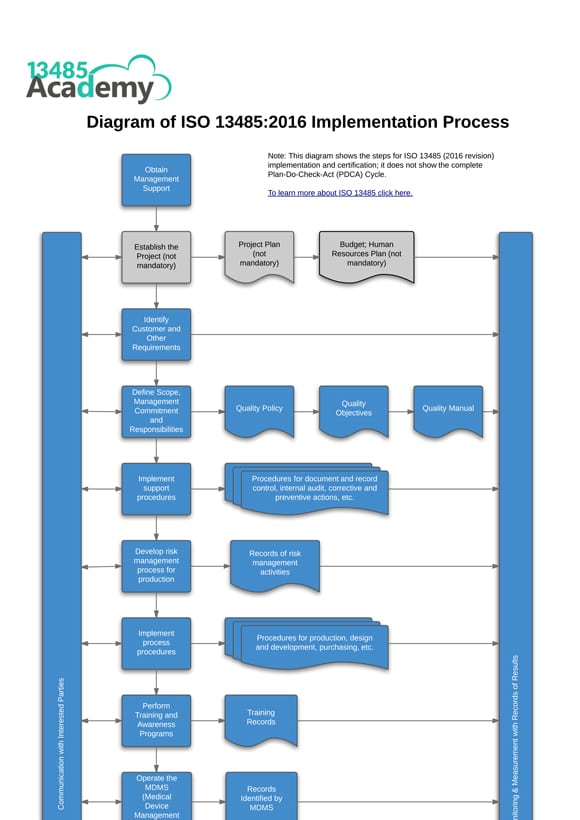
ISO 13485 Quality Management Systems Products and Services Our products and services include: PharmOut frequently implements Quality Management Systems (QMS) compliant with ISO 13485:2016 and / or FDA QSR 820 in medical device & IVD companies. Medicinal Cannabis Industry Conference Sponsors.Medicinal Cannabis Cultivation & Regulations.
Medicinal Cannabis Conference 2020 – 16/17 November.
WHAT IS ISO 13485 IN PLAIN ENGLISH UPDATE
